Easy-to-use asset management solution for medical equipment
IDR monitors your medical devices according to EU legislation, improves patient safety and helps procurement to create cost savings.
Get started for free and take better care of your assets!

IDR is a modern asset management solution for the healthcare industry
Our software meets the strict requirements of EU legislation regarding the monitoring of medical equipment and devices. With IDR, you increase patient safety and create cost savings. We serve customers in all areas of healthcare.
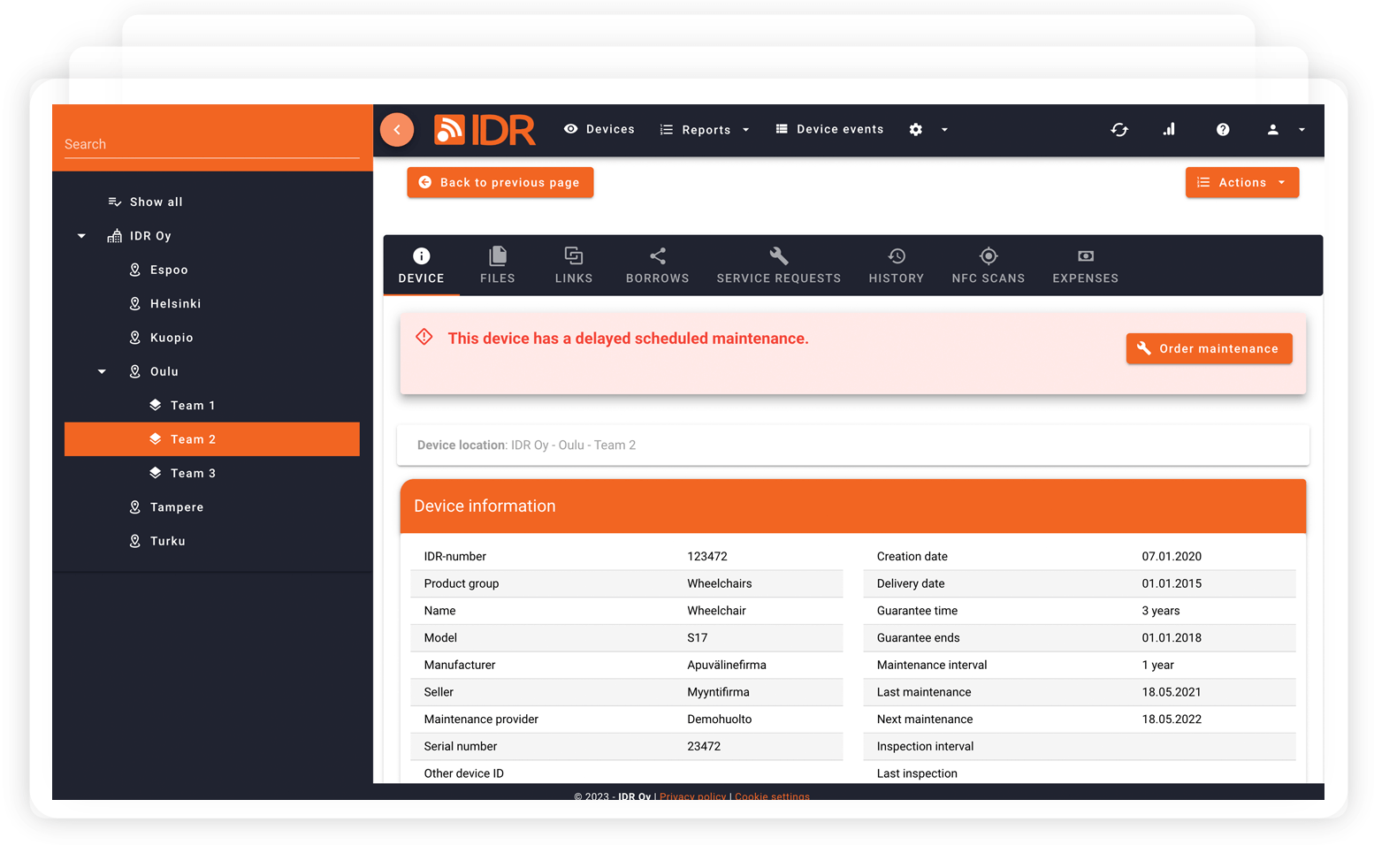
Explore all the features.
What makes IDR the best asset management solution for healthcare?

All important features under one software designed to smooth asset management in healthcare.

All different devices and equipment in one place (online, offline, owned and leased devices).

Link all your maintenance partners into your asset management and forget about shattered operation.

Automate routines with event-based triggers

NFC smart tags for device inventory, service calls, and movement tracking

Own dedicated customer manager for implementation and ongoing support
We help large group of private healthcare organizations and hospitals

“Our mission is to provide a safe home and good care for our residents. To improve patient safety, we have implemented the IDR device registry as a medical device monitoring system in all Esperi units.”
Eeva Ketola
Quality Manager


“The implementation of IDR will ease the professional’s work for years to come. The support for the implementation of IDR has been excellent.”
Anne Komulainen
Chief Dental Officer


“The onboarding process went according to plan from the beginning, and despite the size of the project, everything was completed in three months. Their support was key to the success of the project. We are extremely satisfied with the service and software we have received.”
Jukka Perälä
Medical Director

Automate medical equipment maintenance workflows
IDR helps to automate routines and provides a tailored asset management solution for the industry. With IDR you can easily solve common challenges and take better care of your medical equipment.

Problem
Medical equipment are listed only in Excel and you don’t meet legal requirements. Reporting is non-existent.

Problem
Maintenance of the medical devices is done manually without automation and notifications.

The device inventory is unclear from both quality and procurement perspectives.
Problem
Medical devices are forgotten or used inefficiently. Unnecessary purchases are made.
Read more about the solution.
Read more about asset management in healthcare
Guest blog: Quality and Safety Go Hand in Hand
Guest blog: Quality and Safety Go Hand in HandDeviations are a gift to quality improvement and should not be feared. Instead, it would be surprising if there were no deviations, as who among us is without fault? In our social services and healthcare industry, ensuring...
Improving patient experience through effective asset management in healthcare
In this blog text, we will explore the top six reasons why asset management is vital in the healthcare industry.
How to Choose the Best Asset Management Software for Healthcare Equipment?
In this blog text, we will explore the top six reasons why asset management is vital in the healthcare industry.
Take the first step!
No commitment. No charge. We will present our solution and you can decide if you want to continue with us.
© Copyright – IDR Oy – Privacy Policy



